Williams-Beuren Syndrome: Clinical Features and Genetic Diagnosis Guide
Williams-Beuren Syndrome is a rare neurodevelopmental disorder caused by a 1.5–1.8 Mb hemizygous deletion at chromosome 7q11.23, occurring in approximately 1 in 7,500–10,000 live births worldwide.
📍Key Takeaways
- Williams-Beuren Syndrome results from a hemizygous 7q11.23 deletion; supravalvular aortic stenosis (SVAS) is the hallmark cardiovascular finding, present in approximately 75% of patients.
- Standard G-banded karyotyping cannot detect the 7q11.23 deletion; WGS or WES enables simultaneous confirmation of the deletion and identification of co-occurring variants in a single test.
- WGS demonstrates an overall diagnostic yield approximately 3.4% higher than CMA (Chau et al., 2020) and is the preferred approach for atypical presentations or CMA-negative cases with strong clinical suspicion.

1. Etiology: 7q11.23 Deletion and Genetic Mechanism
WBS results from a hemizygous deletion of approximately 26–28 genes at chromosome 7q11.23, including ELN (elastin), LIMK1, and GTF2I. Haploinsufficiency of ELN is the primary driver of the cardiovascular phenotype, particularly supravalvular aortic stenosis (SVAS).¹
- Mechanism: Over 95% of deletions arise through non-allelic homologous recombination (NAHR) mediated by flanking low-copy repeats (LCRs).²
- Inheritance: The vast majority (>95%) of cases are de novo; familial transmission accounts for fewer than 5% of cases.
2. Key Clinical Features
WBS presents as a multisystem disorder affecting cardiovascular, craniofacial, neurocognitive, and connective tissue systems.
Cardiovascular Abnormalities (prevalence ~75–80%)³
- SVAS is the most prevalent cardiac finding, identified in approximately 75% of patients.
- Peripheral pulmonary stenosis, aortic coarctation, and other vascular stenoses may co-occur.
Craniofacial Features
- Characteristic elfin facies: broad forehead, periorbital fullness, depressed nasal bridge, long philtrum, full lips
- Stellate iris pattern: observed in approximately 74% of patients⁴
Neurocognitive and Behavioral Profile
- Mild-to-moderate intellectual disability (mean IQ ~56)
- Relative preservation of verbal abilities with marked visuospatial deficits
- Hypersociability, specific phobias, and elevated rates of anxiety disorders
Other Features
- Infantile hypercalcemia: present in approximately 15% of cases
- Growth restriction: mean adult height ~161 cm (males), ~152 cm (females)
💡Williams-Beuren Syndrome in Clinical Practice
As of March 2026, approximately 40 patients have been confirmed with Williams-Beuren Syndrome through 3billion’s WES/WGS testing — all confirmed via 7q11.23 CNV (deletion).
The most striking finding is the gap between the referral phenotype and the textbook presentation. Supravalvular aortic stenosis (SVAS), widely described in the literature as the hallmark feature of WBS, was recorded as the primary presenting symptom in only approximately 15% of cases in our real-world data. The findings that actually drove referral were neurodevelopmental delay (~56%) and facial dysmorphism (~36%) — suggesting that in clinical practice, patients with WBS are far more likely to reach genetic testing through “developmental delay and dysmorphic features” than through “cardiac abnormalities.”
Growth restriction (~28%) and hearing anomalies (~28%) were also present at notable rates, alongside hypotonia (~23%) and ocular abnormalities (~23%). While the neonatal and infantile periods were the most common time of onset, a substantial number of cases had symptoms first recorded in childhood — highlighting the potential for diagnostic delay.
Ultimately, the real-world data leaves one clinical question: in a patient with developmental delay and facial dysmorphism but no SVAS, is Williams-Beuren Syndrome on your differential?
3. Diagnostic Criteria and Clinical Assessment
WBS diagnosis is based on a combination of characteristic phenotypic features. Initial clinical workup should include:
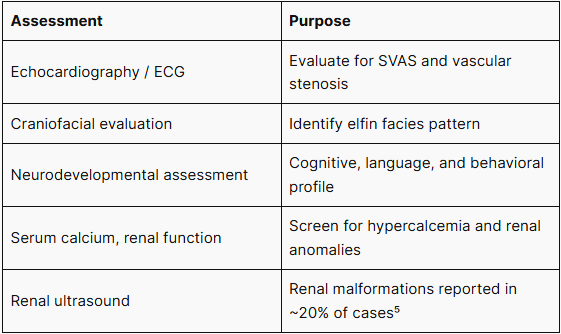
Clinical findings alone are insufficient for a definitive diagnosis; molecular genetic confirmation is mandatory.
4. Genetic Testing Workflow for Diagnosis
When to Order Genetic Testing

Genetic testing for WBS should be considered when two or more of the following are present:
- SVAS or peripheral pulmonary stenosis in infancy
- Characteristic elfin facies
- Language delay combined with hypersociable behavior
- Unexplained infantile hypercalcemia
Recommended Testing Approaches
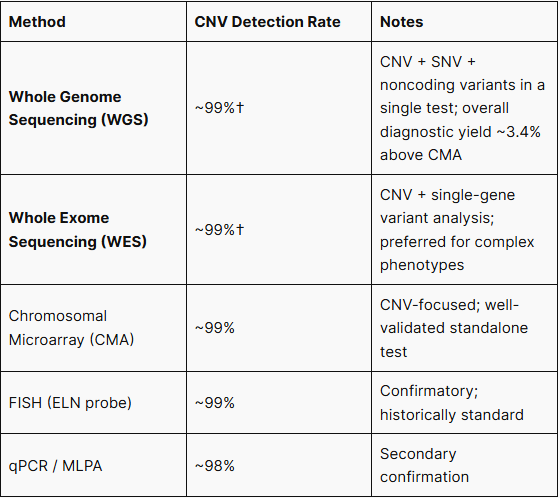
WGS and WES detect the 7q11.23 deletion with accuracy equivalent to CMA, while simultaneously capturing co-occurring SNVs, phenotype-modifying rare variants, and noncoding region changes — all from a single test. In a head-to-head comparison of 532 clinical cases, low-pass WGS achieved an overall diagnostic yield of 23.1% versus 22.4% for CMA, a relative improvement of approximately 3.4%.⁶ A separate validation study confirmed 100% concordance between LP-WGS and CMA across 55 CNVs ranging from 75 kb to 90.3 Mb.⁷ For patients with atypical presentations, CMA-negative results despite strong clinical suspicion, or where concurrent evaluation of comorbid genetic conditions is warranted, WGS or WES is the preferred comprehensive diagnostic approach.⁸
Chau et al., Hum Genet. 2020; Mazzonetto et al., Ann Hum Genet. 2023
5. Prognosis and Genetic Counseling
Life expectancy in WBS is reduced compared to the general population, primarily due to cardiovascular complications. Recurrence risk is 50% for affected individuals and approximately 1% for unaffected parents with a de novo deletion in a child.⁹ Prenatal diagnosis via CMA or FISH on chorionic villus or amniocentesis samples is available.
FAQ
Q. Can standard karyotyping detect Williams-Beuren Syndrome? No. The 7q11.23 deletion is below the resolution of conventional G-banded karyotyping. FISH or chromosomal microarray testing is required for diagnosis.
Q. Is prenatal diagnosis possible? Yes. CMA or FISH performed on fetal cells obtained via amniocentesis or chorionic villus sampling (CVS) allows prenatal diagnosis.
References
- Merla G, et al. Hum Mol Genet. 2010;19(R2):R174–R179.
- Bayes M, et al. Am J Hum Genet. 2003;73(1):131–151.
- Pober BR. N Engl J Med. 2010;362(3):239–252.
- Winter M, et al. Eur J Pediatr. 1996;155(12):1037–1042.
- Pankau R, et al. Am J Med Genet. 1996;63(2):301–306.
- Chau MHK, et al. Hum Genet. 2020;139(11):1403–1415.
- Mazzonetto PC, et al. Ann Hum Genet. 2024;88(2):113–125.
- Manning M, Hudgins L. Genet Med. 2010;12(12):742–745.
- Metcalfe K, et al. J Med Genet. 2005;42(2):e9.
If you’re considering genetic testing for a patient with suspected Williams-Beuren Syndrome, our clinical team is ready to support your diagnostic workup.
Get exclusive rare disease updates
from 3billion.

Soo-jung Baek
As a marketer, I strive to empower the rare disease community by sharing meaningful insights backed by our company’s expertise.