Treacher Collins Syndrome: Identifying Genetic Causes via Advanced Sequencing
📍Key Takeaways
1. Resolving Genetic Heterogeneity and Inheritance Uncertainty TCS is caused by mutations in TCOF1, POLR1B, POLR1C, or POLR1D. Genetic testing is essential to differentiate between autosomal dominant and recessive inheritance, resolving uncertainties that clinical observation alone cannot address.
2. Enhancing Accuracy in Recurrence Risk Counseling Misclassifying the inheritance pattern based solely on phenotype can lead to significant errors in recurrence risk counseling (25% vs. 50%). Molecular diagnosis provides the definitive evidence needed for accurate genetic counseling and family planning.
3. Establishing Standards for Multidisciplinary Care and Early Intervention From neonatal airway management to early hearing intervention and staged craniofacial reconstruction, early confirmation via genetic testing serves as a milestone for determining the timing of multidisciplinary interventions and managing patient prognosis.

Treacher Collins Syndrome (TCS) is a rare craniofacial disorder whose diagnostic complexity extends well beyond its visible phenotype. Relying solely on clinical features without molecular confirmation risks misclassifying inheritance patterns — a consequential error when counseling families on recurrence risk. This guide provides clinicians and genetic counselors with an evidence-based framework for understanding TCS genetics and ordering appropriate genetic tests.
1. What Is Treacher Collins Syndrome?
TCS (OMIM #154500) occurs in approximately 1 in 10,000 to 1 in 50,000 live births and is characterized by bilateral, symmetrical craniofacial malformations affecting structures derived from the first and second pharyngeal arches. It is inherited predominantly in an autosomal dominant manner. Intelligence is typically preserved, though phenotypic severity varies considerably — even within the same family.
2. Genetic Basis: TCOF1, POLR1C, POLR1D, and POLR1B
The genetic basis of TCS involves four primary genes:
- TCOF1 (5q32-q33.1): Accounts for approximately 88% of TCS cases. It encodes the Treacle protein, which localizes to the nucleolus and regulates ribosomal RNA biogenesis. Loss of function impairs neural crest cell (NCC) proliferation and survival during craniofacial development. Inheritance is autosomal dominant.
- POLR1C (6p21.1) and POLR1D (13q12.2): These encode subunits shared by RNA Polymerases I and III. Critically, POLR1C follows an autosomal recessive pattern (biallelic mutations required), while POLR1D can be either dominant or recessive. Misclassifying a POLR1C-related TCS case as dominant leads to a critical counseling error: 25% vs. 50% recurrence risk.
- POLR1B (2p11.2): Designated TCS type 4, this is the most recently identified TCS gene and follows autosomal dominant inheritance.
3. Clinical Features and Phenotypic Variability
Core clinical features include:
- Malar and mandibular hypoplasia: Zygomatic arch hypoplasia with characteristic downslanting palpebral fissures.
- Ear anomalies: Microtia/anotia and external auditory canal atresia, causing conductive hearing loss in many affected individuals.
- Eyelid defects: Lower eyelid coloboma with absence of medial eyelashes.
- Cleft palate: Present in approximately one-quarter of patients.
- Airway compromise: Micrognathia can cause life-threatening neonatal airway obstruction.
Intelligence is typically normal. Early audiological intervention — including bone-anchored hearing aids — is critical for language and cognitive development.
4. Inheritance and Recurrence Risk

- Autosomal Dominant (TCOF1/POLR1B): Recurrence risk is 50% for offspring of an affected parent. However, a substantial proportion of TCS cases arise de novo, so absence of family history does not exclude the diagnosis. Families with de novo variants should be counseled about the rare possibility of germline mosaicism.
- Autosomal Recessive (POLR1C): Carries a 25% recurrence risk per pregnancy. This underscores the critical importance of gene-specific molecular confirmation before counseling.
5. Diagnostic Approach: When and How to Order Genetic Testing
The recommended testing strategy is as follows:
- Multi-gene craniofacial/skeletal panel: Simultaneous analysis of TCOF1, POLR1C, POLR1D, and POLR1B — recommended as the first-line test.
- WES/WGS: Consider when the panel is negative or the phenotype is atypical.
- CNV analysis (MLPA/array CGH): Large TCOF1 deletions and duplications are missed by sequencing alone; MLPA is recommended as an adjunct.
6. Prenatal Diagnosis and Genetic Counseling
For families with a known pathogenic variant, prenatal molecular testing via CVS (10–12 weeks) or amniocentesis (15–20 weeks) is available. When fetal ultrasound reveals craniofacial anomalies without prior family history, fetal WES should be considered to include TCS in the differential diagnosis.
7. Multidisciplinary Management
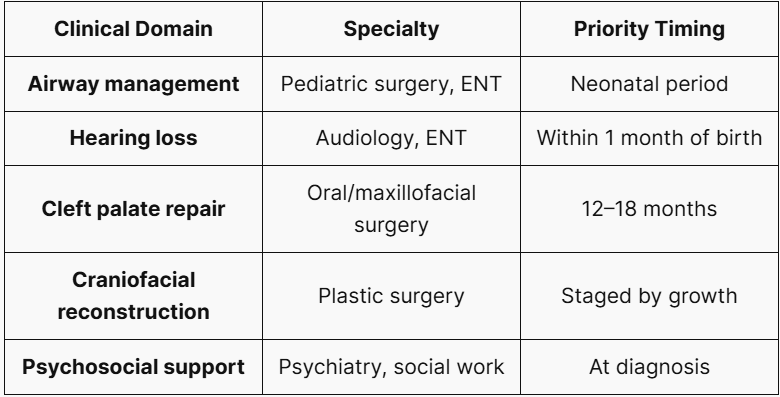
Because the inheritance patterns and recurrence risks of Treacher Collins Syndrome (TCS) vary significantly depending on the underlying causative gene, molecular confirmation is essential to complete the clinical diagnosis. Moving beyond the limitations of single-gene tests or small targeted panels, 3billion provides optimized diagnostic solutions through data-driven Whole Exome Sequencing (WES) and Whole Genome Sequencing (WGS).
If you are looking to provide the most accurate genetic answers for patients with suspected Treacher Collins Syndrome, explore 3billion’s advanced whole-genome analysis services.
References
- Dixon J, Trainor P, Dixon MJ. Treacher Collins syndrome. Orthod Craniofac Res. 2007. https://doi.org/10.1111/j.1601-6343.2007.00388.x
- Dauwerse JG, et al. Mutations in genes encoding subunits of RNA polymerases I and III cause Treacher Collins syndrome. Nat Genet. 2011. https://doi.org/10.1038/ng.724
- Sanchez E, et al. POLR1B and neural crest cell anomalies in Treacher Collins syndrome type 4. Genet Med. 2020. https://doi.org/10.1038/s41436-019-0669-9
- Ulhaq ZS, et al. A systematic review on Treacher Collins syndrome. Clin Genet. 2023. https://doi.org/10.1111/cge.14243
- Li X, et al. Genotype-phenotype variability in Chinese cases of Treacher Collins syndrome. Acta Otolaryngol. 2019. https://doi.org/10.1080/00016489.2019.1612530
Get exclusive rare disease updates
from 3billion.

Soo-jung Baek
As a marketer, I strive to empower the rare disease community by sharing meaningful insights backed by our company’s expertise.