Which Children with Motor Speech Disorders Should Be Prioritised for Genetic Testing?

At a Glance
When delayed walking (OR 15.96), receptive language impairment, borderline IQ, dysmorphic features, or motor delays are present, exome sequencing should be considered first.A landmark clinical study has finally answered a question families have been asking for years.
The clinical genetic diagnostic yield for childhood motor speech disorders (CAS/dysarthria) is 29% — on par with epilepsy and cerebral palsy.
More Common Than You Might Think
Speech difficulties are one of the most frequent concerns that bring children to paediatric and primary care clinics. Around 5% of all children experience some form of speech sound disorder — articulation problems, phonological delays, or unclear pronunciation. For most of them, the outlook is reassuring: these conditions typically resolve by around age seven, with or without speech therapy.
But within that group, there is a smaller and far more challenging subset.
Some children don’t get better. Their speech remains inconsistent, difficult to understand, and resistant to therapy — often well into adolescence or adulthood. These children are estimated to represent approximately 1 in 1,000 in the general population, and they carry a diagnosis of one of two motor speech disorders:
- Childhood Apraxia of Speech (CAS): a disruption in the planning and sequencing of speech movements, resulting in inconsistent errors, impaired coarticulatory transitions, and abnormal prosody. Implicates frontal lobe and basal ganglia pathways.
- Dysarthria: a disorder of neuromuscular control and execution of speech, presenting with abnormal oral tone, imprecise articulation, and hypernasality. Linked to dysfunction of the corticobulbar tract and the “final common” motor pathway.
Both conditions can co-occur, and when they do, the clinical picture becomes more complex.
Families Without Answers
For the families of these children, one of the most difficult realities is simply not knowing why.
Normal MRI. Normal metabolic workup. Years spent moving between paediatrics, neurology, and rehabilitation — without a clear explanation. This experience has a name: the diagnostic odyssey.
Systematic research into the genetic causes of motor speech disorders only began in earnest over the past five years. Research cohorts began reporting diagnostic yields of around 30%, but no study had examined what that yield looks like when clinical genetic testing is applied in a real hospital setting — until now.
The First Clinical Measurement of Genetic Diagnostic Yield
A study published in January 2026 in the European Journal of Human Genetics (Van Niel et al.) filled this gap for the first time.
At a tertiary paediatric speech genomics clinic at the Royal Children’s Hospital in Melbourne, Australia, 153 children (aged 2y7ms to 16y5ms) confirmed to have CAS or dysarthria underwent a clinically accredited genomic testing pipeline. The workup consisted of three components — and across the cohort, a genetic diagnosis was identified in approximately 1 in 3 children.
- CMA (Chromosomal Microarray): detection of large chromosomal deletions and duplications
- Fragile X PCR: screening for Fragile X syndrome
- Exome Sequencing: single-gene variant analysis (trio testing with parents where possible)
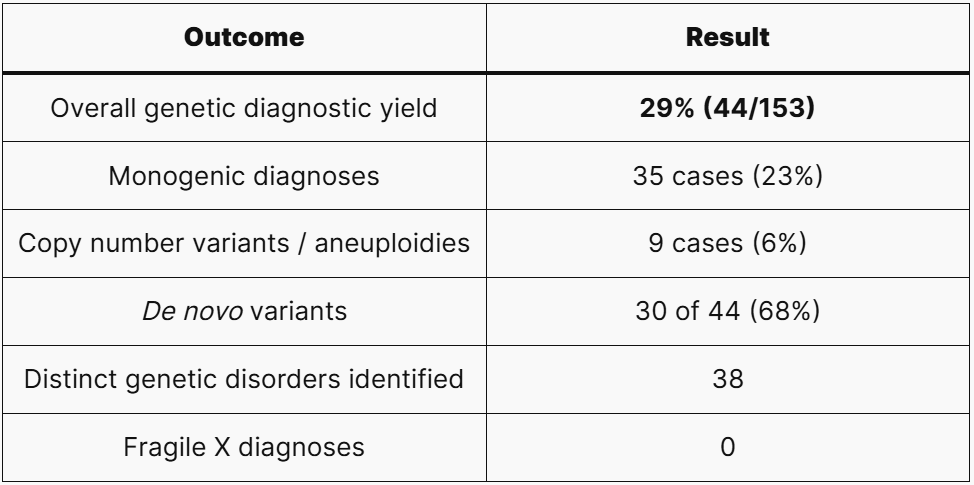
Nearly one in three children received a confirmed genetic diagnosis. The majority of pathogenic variants arose de novo — meaning they were not inherited from either parent, which has direct implications for recurrence counselling.
What Does 29% Actually Mean?
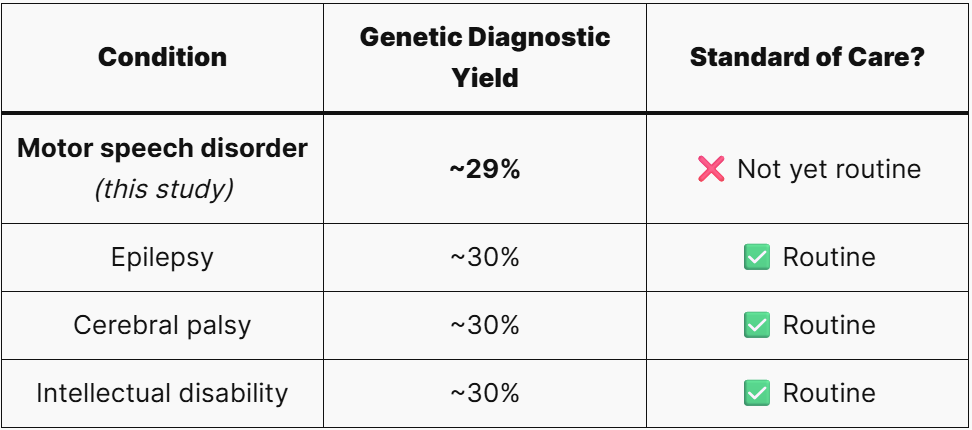
Compared to other neurodevelopmental conditions where genetic testing is already standard of care, the yield is virtually identical.
That’s a striking finding. Children with epilepsy or cerebral palsy are routinely referred for genetic testing. Children with motor speech disorders still are not. This study calls that out as a problem.
Which Genes Were Implicated?
Across the 44 children who received a diagnosis, 38 distinct genetic The pathogenic variants identified fell into three broad groups:
- Chromatin regulators and transcriptional modulators: the largest group, with SET domain proteins — SETD1A, SETD2, SETD5, NSD1, EHMT1 — appearing repeatedly. These are members of the methyltransferase superfamily with a well-established role in post-translational protein regulation and neurodevelopmental disorders.
- Ion channels and solute carriers: including CACNA1A, SCN8A, KCND3, SLC6A1, and SLC6A8.
- Cell signalling pathway members: including FBXW7, RAF1, PPP2R5D, and TAB2.
Notably, 15 genes — including ADGRL1, ANK2, BPTF, CAMK2A, CUX1, FBXW7, KCND3, NSD1, RAF1, SETD2, SLC6A8, SPTBN1, SRRM2, TAB2, and TRIM8 — were reported for the first time as causes of CAS and dysarthria.
Who Should Be Tested First?
The research team analysed clinical variables to identify which features predicted a genetic diagnosis. The odds ratios were telling.
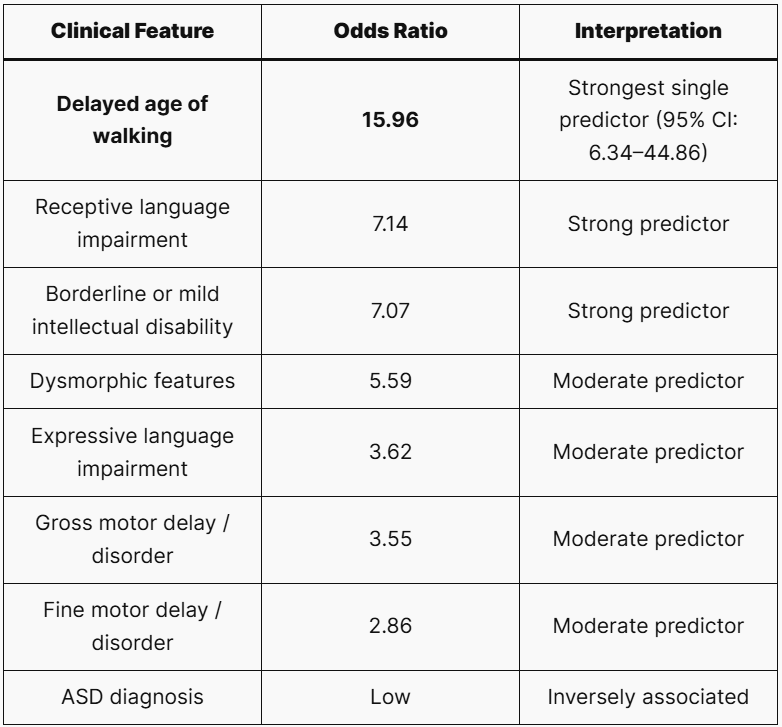
Delayed walking stood out as the strongest single predictor (OR 15.96). In a busy outpatient clinic, asking “When did your child first walk?” can be one of the most diagnostically meaningful questions you ask.
Co-occurring CAS and dysarthria also raised the probability significantly — more so than CAS alone. When both are present, the threshold for requesting testing should be lower.
Among children who did not receive a genetic diagnosis, 47.7% had ASD features. This suggests that motor speech disorders in this subgroup may follow a polygenic rather than monogenic architecture — similar to the genetic landscape of autism itself. Children where ASD is the primary co-occurring feature, without other red flags, can reasonably be deprioritised for genetic testing.
Finally, Fragile X PCR returned zero diagnoses in this cohort — consistent with all prior prospective CAS studies. While one study is not sufficient to draw firm conclusions, the utility of Fragile X testing as a first-line screen in children presenting primarily with motor speech disorders warrants reconsideration.
Summary: When to Consider Genetic Testing First
In a child with a primary motor speech disorder (CAS and/or dysarthria), clinical genomic testing should be actively considered when any of the following are present:
✅ Delayed age of walking — the strongest single signal
✅ Receptive language impairment
✅ Borderline or mild intellectual disability
✅ Co-occurring CAS and dysarthria
Conversely, when ASD is the dominant co-occurring feature and none of the above apply, the likelihood of a monogenic diagnosis is lower, and these children can be deprioritised for routine genetic referral.
The recommended workup is exome sequencing (trio where possible), with chromosomal microarray added as needed.
Closing Thoughtsord
Many children present with delayed speech. But within that group, there are children for whom the question “why?” has a real, findable answer. This study — the first to measure clinical genetic diagnostic yield in motor speech disorders — makes the case that these children deserve the same diagnostic rigour we apply to epilepsy and cerebral palsy.
A single genetic result can end years of uncertainty. It can give a family a diagnosis, a direction, and a path toward precision-based care.
It starts with one question in clinic: “When did your child first walk?”
3billion provides exome sequencing supported by automated AI-driven reanalysis. If you have questions about the testing process, sample collection, or how to get started, our team is ready to help.
This article is intended for healthcare professional education and information sharing. Clinical decisions for individual patients should always be made in consultation with the treating medical team.
Get exclusive rare disease updates
from 3billion.

Sookjin Lee
Expert in integrating cutting-edge genomic healthcare technologies with market needs. With 15+ years of experience, driving impactful changes in global healthcare.






