[Clinical Review] Why WGS Achieves up to 44.4% Diagnostic Yield in NDD: Insights from a 100-Family Italian Study
🎯 Key Highlights
Prioritization of WGS: For patients with Neurodevelopmental Disorders (NDDs)—particularly those with comorbid Intellectual Disability (ID)—Whole Genome Sequencing (WGS) serves as the most powerful first-tier diagnostic tool, providing an approximate 10% increase in diagnostic yield over conventional testing methods.
Necessity of Trio Analysis: Precise interpretation through Trio-based analysis is essential, as complex variants inherited from parents with subclinical or mild symptoms can manifest as severe phenotypes in their offspring.
Value of Secondary Findings: Beyond the primary NDD diagnosis, WGS offers significant preventive medical value by uncovering clinically actionable genetic risks in genes such as OTC, BRCA2, and MYBPC3, which require immediate clinical management.

When assessing patients with neurodevelopmental disorders (NDDs) in the clinic, you have likely encountered the challenges of overlapping symptoms and blurred boundaries between Autism Spectrum Disorder (ASD) and Intellectual Disability (ID). If you have ever hesitated over whether to pursue deeper genetic testing for a patient with a complex, unexplained phenotype, a recent study published in npj Genomic Medicine offers a compelling clinical answer.
This study analyzed the whole genomes of 110 probands (from 100 families, totaling 298 individuals) affected by NDDs in the Valle d’Aosta region of Italy.
From a clinician’s perspective, here are three key takeaways on why this study is significant—and why you should consider WGS for your own undiagnosed patients.
1. Striking Differences in Diagnostic Yield Across Stratified Clinical Groups
Because the NDD spectrum is highly heterogeneous, researchers stratified the probands into three distinct clinical categories:
• ASD only: Autism spectrum disorder without intellectual disability (primarily high-functioning cases).
• ASD-ID: ASD with comorbid intellectual disability and/or severe language impairment.
• ID only: Intellectual disability without ASD.
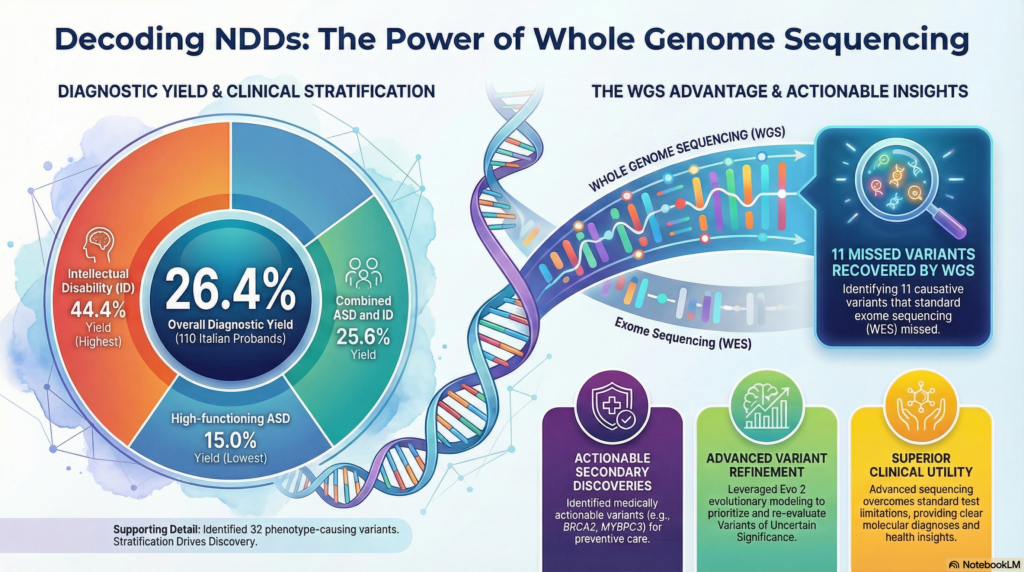
Overall, the study identified likely phenotype-causing variants in 26.4% of the probands. More interestingly, the diagnostic yield showed a clear gradient based on the clinical subgroups. The diagnostic yield for the ID-only group reached a remarkable 44.4%, demonstrating the decisive role of comprehensive genetic testing in these patients.
Perhaps more surprisingly, even within the high-functioning ASD-only group—a condition often presumed to be heavily influenced by polygenic or environmental factors—15% of patients harbored a disease-causing single-gene variant. This provides strong clinical evidence against the notion that extensive genetic testing is unnecessary for patients with high-functioning autism, proving that proactive screening can uncover hidden pathogenic variants.
2. The Power of WGS: Overcoming the Limitations of Whole Exome Sequencing (WES)
While Whole Exome Sequencing (WES) is currently the most cost-effective method in clinical diagnostics, it leaves approximately 70% of diagnosed NDD individuals unsolved.
This study provides strong evidence for why a gradual transition to WGS is necessary. Of the 32 disease-causing variants identified, 11 would have been completely missed by conventional WES. WGS comprehensively detects variants in non-coding regions, copy-number variants (CNVs), and structural variants (SVs), effectively increasing the diagnostic yield by approximately 10%.
A Complex Familial Case (Family NED029): In one family, both parents exhibited only mild behavioral traits without a formal diagnosis, while the proband suffered from severe ASD and ID. Conventional targeted testing would likely have failed to find the cause. However, through trio-based WGS, researchers discovered that the proband had inherited a complex combination of micro-structural variants (CNVs) in multiple neurodevelopment-related genes (e.g., KMT2C, GLRA1, NPAS1) from both parents. This perfectly illustrates WGS’s unique ability to piece together complex, polygenic, or multifactorial genetic puzzles.
3. AI-Driven Re-evaluation and Life-Saving ‘Secondary Findings’ for Families
Variants of uncertain significance (VUS) are a common source of clinical frustration. In this study, researchers utilized Evo 2, an AI-based evolutionary constraint model, to refine VUS interpretation. The algorithm revealed that 8 VUS in genes not previously strongly linked to ASD (such as KALRN) exhibited pathogenic-like signatures, opening the door to new diagnostic criteria and novel candidate genes.
Furthermore, WGS can identify medically actionable secondary findings that could save the lives of patients and their families. Even though the primary indication was an NDD diagnosis, the study uncovered pathogenic variants in genes like MYBPC3 (hypertrophic cardiomyopathy) and BRCA2 (hereditary breast and ovarian cancer syndrome) in the mothers of the probands, enabling proactive, preventive healthcare.
Could that undiagnosed patient in your clinic be missing a hidden genetic puzzle piece?
Because ASD and ID frequently co-occur and share complex phenotypic traits, isolating the underlying etiology through clinical evaluation alone is incredibly difficult. Although barriers such as diagnostic cost and analytical complexity remain, this study clearly demonstrates that Whole Genome Sequencing (WGS) acts as a superior diagnostic tool, offering new hope for unsolved NDD cases—especially those who previously tested negative on WES or present with complex comorbidities.
Today, you might want to reopen the charts of those patients whose prolonged diagnostic odysseys have caused anxiety for their families, and strongly consider adding WGS to your diagnostic arsenal.
Get exclusive rare disease updates
from 3billion.

Sookjin Lee
Expert in integrating cutting-edge genomic healthcare technologies with market needs. With 15+ years of experience, driving impactful changes in global healthcare.